Glucose.
Glucose is the most mutual monosaccharide. It is known equally Dextrose because it occurs inwards nature principally equally the optically active dextrorotatory isomer. Glucose is found inwards most sweetness fruits, peculiarly grapes (20-30%), in addition to honey. It is an essential component subdivision of human blood. The blood ordinarily contains 65 to 110 mg (0.06 to 0.1%) of glucose per 100 ml. inwards diabetic persons, the degree may live much higher. In the combined shape glucose occurs inwards abundance inwards cane refined saccharify in addition to polysaccharides such equally starch in addition to cellulose.
Preparation of glucose.
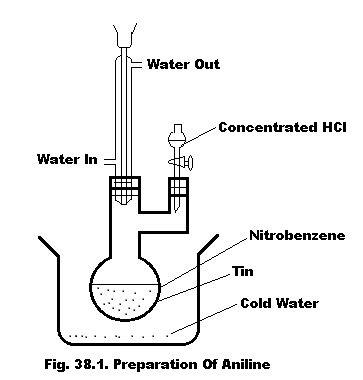
Glucose is produced commercially past times the hydrolysis of starch amongst dilute muriatic acid at high temperature nether pressure.
An aqueous interruption of starch obtained from corn is acidified amongst hydrochloric acid, It is so heated amongst high-pressure steam inwards an autoclave. When the hydrolysis is complete, the liquid is neutralized amongst sodium carbonate to a pH of 4-5. The resulting solution is concentrated nether reduced pressure level to larn the crystals of glucose.
Uses of glucose.
- As a sweetening agent inwards syrups in addition to confectionery.
- As nutrient for infants.
- As a reducing agent inwards the silvering of mirrors in addition to to convert indigo blueish to indigo white inwards vat dyeing.
- As a raw textile for vino in addition to alcohol manufacture.
Structure of glucose.
The structure of glucose may live discussed nether the next heads.
- Open Chain formula.
- Configuration.
- Cyclic structure.
- Haworth representation.
Open-chain Formula.
The open-chain formula of glucose is constructed from the next facts:
Elemental analysis in addition to molecular weight decision accept established that glucose has the molecular formula C6H12O6.
(2) Presence of 6-carbon unbranched chain.
The consummate reduction of glucose amongst concentrated hydrogen iodide in addition to crimson phosphorus gives n-hexane. This proves that a glucose molecule is made of an unbranched six-carbon chain.
(3) Presence of 5 OH group.
Glucose reacts amongst acetic anhydride to shape the pentadactyl derivative. This shows the presence of v hydroxy groups. Since glucose is a stable compound, no 2 OH groups are attached to the same carbon. In other words, the v OH groups are on unlike carbons.
(4) Presence of the C=O group.
Glucose reacts amongst hydroxylamine to shape an oxime. It suggests the presence of a carbonyl group.
(5) Presence of terminal CHO function.
On mild oxidation amongst bromine water, glucose is converted to glucose acid which when reduced amongst an excess of Hl yields n-hexanoic acid.
This shows that glucose contains a six-carbon conduct chain amongst CHO at i end, which has been oxidized to COOH.
(6) Construction of open-chain formula.
Knowing that glucose has a conduct 6-carbon chain amongst a terminal CHO, the v OH groups tin sack live placed i each on the remaining v carbons. Supplying hydrogen atoms to these carbons to satisfy their tetracovalency, the open-chain construction of glucose tin sack live written equally :
Configuration of D-Glucose.
The configuration of D-glucose was proved past times Emil Fischer past times arguments similar to the ones stated below.
(1) Construction of 4 possible D-pentoses.
Taking the configuration of D-glyceraldehyde equally the standard, 2 possible D-aldotetroses (A in addition to B) may live constructed past times adding a CHOH simply below CHO, placing OH to the right in addition to so to the left.
Similarly, each of the 2 D-tetroses (A in addition to B) gives 2 D-aldopentoses. Thus 4 possible D-aldopentoses are :
(2) D-Arabinose has configuration II or IV.
Oxidation of D-arabinose amongst nitric acid oxidizes the terminal CHO in addition to CH2OH groups yielding 2 optically active (asymmetric) dicarboxylic acids. The forms II in addition to IV tin sack shape 2 optically active (asymmetric) diacids, land I in addition to III tin sack give meso acids solely that has a bird of symmetry. Therefore, D-arabinose is either II or IV.
(3) Configuration II confirmed for D-arabinose.
D-arabinose past times Killiani -Fischer synthesis yields 2 epimeric aldohexoses, D-glucose in addition to D-mannose. These on oxidation amongst nitric acid shape 2 optically active (asymmetric) dicarboxylic acids. This is theoretically possible solely if D-arabinose has the configuration II in addition to non IV.
Proceeding similarly, yous volition detect that if D arabinose had configuration IV, of the 2 dicarboxylic acids derived from it, i would live meso in addition to i asymmetric (optically active). Hence D-arabinose has configuration II.
(4) Ruff degradation of D-glucose in addition to D-mannose produces D- arabinose inwards each case.
In ruff degradation, the CHOH below CHOH is destroyed. Therefore, the configuration of the 2 aldohexoses, D-glucose in addition to D-mannose, tin sack live derived past times adding a novel CHOH below CHO inwards shape II of D-arabinose.
Hence D-glucose has configuration V or VI.
(5) D-glucose in addition to L-glucose yield the same dicarboxylic acid.
This way that these 2 sugars differ solely inwards abide by of the pose of the terminal groups (CHO in addition to CH2OH). Therefore, the telephone substitution of the terminal groups inwards D-glucose should live able to give a unlike aldohexose (L-glucose). Let us instantly examine configuration formulas V in addition to VI (One of which is D-glucose) from this angle.
If VII is rotated through 180° in the bird of the paper, it gives an aldohexose VIII, unlike from V. Influenza A virus subtype H5N1 similar physical care for amongst formula VI does non give rising to a unlike sugar.
From the inwards a higher house arguments, it is evident that D-glucose has the configuration equally shown past times the from V.
Cyclic Structure of D-Glucose.
(1) Open-Chain Structure non wholly true.
Fischer realized that the open-chain pentahydroxy aldehyde construction of glucose did non wholly explicate its chemic behavior. Unlike unproblematic aldehydes, glucose did non shape the crystalline bisulfite chemical compound in addition to failed to give the shiff's test. Furthermore, the penta-acetate in addition to pentamethyl-ether derivatives of glucose are non oxidized past times Tollens' regent or Fehling's solution, indicating the absence of CHO group.
(2) The cyclic construction suggested explaining mutarotation.
The French pharmacist Tarnet established the beingness of 2 crystalline forms of glucose, α-glucose, and β-glucose. α-Glucose had specific rotation +112°, while β-glucose + 19°. The optical rotation of each of these forms changed gradually amongst fourth dimension till survive a constant value of +53° was reached. To explicate this phenomenon of mutarotation, it was visualized that the α and β glucose were, inwards reality, the cyclic hemiacetal forms of glucose which were interconvertible via the open-chain form. The constant value of + 19° represented the Earth of equilibrium between α-D-glucose and β-D-glucose.
The concentrations of α-D glucose, β-D-glucose, in addition to the open-chain glucose at equilibrium are 36%, 64%, in addition to less than 0.01% respectively. This explains why D-glucose tin sack react both equally an aldehyde in addition to a cyclic hemiacetal inwards which CHO is absent.
(3) Glycoside formation confirms the cyclic structure.
Glucose when treated amongst methanol inwards the presence of dry out HCl, gives 2 isomeric acetals or glycosides,. These crystalline glycosides, methyl-α-D-glucose, in addition to methyl-β-D glucoside accept been genuinely isolated. These are optically active but make non give whatever reactions to the gratuitous CHO group. Evidently, the 2 glycosides are the methyl derivatives of α- and β-D-glucose, formed equally a number of the reaction betwixt the hemiacetal OH of these forms in addition to methanol.
thus the cyclic construction of D-glucose stands confirmed but whether it has a 5-membered or a 6-membered band is nonetheless to live proved.
(4) Determination of band size.
So far nosotros accept represented the construction of cyclic hemiacetals or anomers of D-glucose equally having a band of vi members, v carbons in addition to i oxygen. This has been proved to live right in addition to a five-membered band has been ruled out.
Hirst (1926) prepared tetra-O-methyl-D-glucose. past times treating methyl-D-glucoside amongst dimethyl sulfate in addition to subsequent acid hydrolysis of the pentamethyl derivative formed. The oxidation of tetra-O-methyl-D-glucose amongst nitric acid yielded trimethoxyglutaric acid.
Obviously, the 2 carboxylic carbons (1,5) of the trimethoxyglutaric acid are the ones originally involved inwards a band formation. Hence, at that topographic point must accept existed an oxide band betwixt C-1 in addition to C-5. Tracing dorsum the reaction sequence, it stands proved that D-glucose has a six-membered ring. The presence of a 6-membered band inwards D-glucose has too been confirmed past times x-ray analysis.
The Haworth Representation.
So far nosotros accept used Fischer projection formulas for representing the cyclic forms of D-glucose. Haworth idea that these structures were awkward. He introduced the hexagonal representations resembling the heterocycle pyran which contains five-carbon in addition to i oxygen inwards the ring. Thus, he claimed the names α-D-glucopyranose and β-D-glucopyranose for the hexagonal structures of α-D-glucose and β-D-glucose.
It may live noted that inwards Haworth formula, all the OH groups on the right inwards Fischer formula are directed below the bird of the ring, land those on the left larn inwards a higher house the plane. The terminal CH2OH projects inwards a higher house the bird of the ring.
Physical properties of glucose.
Glucose is a white crystalline solid, mp 146°C. When crystallized from mutual coldness water, it forms glucose monohydrate (C6H12O6.H2O), mp 86°C. it is extremely soluble inwards water, solely sparingly so inwards ethanol, in addition to insoluble inwards ether. It is most three-fourths equally sweetness equally cane refined saccharify (sucrose). It is optically active, in addition to the ordinary naturally occurring shape is (+)-glucose.
Chemical properties of glucose.
We accept seen that D-glucose is an equilibrium mixture of a straight-chain shape in addition to a cyclic hemiacetal form.